Updated 1 month ago
EO-Sterilizable Urine Sample Containers: Raising the Bar for Pathogen Transport Safety


Admin
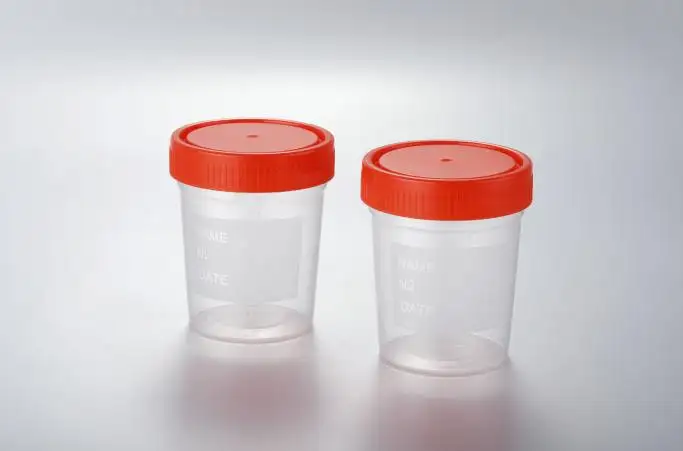
Urine Sample Containers
Recently developed Urine Sample Containers designed modern diagnostics have changed specimen containment practices. Containers safeguard specimen integrity. Patients who present with infectious pathogens (bacteria, viruses and fungi) present additional risks. Containers transform into biosafety devices. When containers are also EO-sterilizable, they protect the user to an even higher degree.

Trustlab EO-sterilizable Urine Sample Containers are the first of their kind. Trustlab's containers protect sample integrity and laboratory and transport personnel. Trustlab's containers allow physicians to safeguard diagnostic accuracy without compromise.
Why EO Sterilization Matters for Pathogen Transport
Pathogen transport safety rests on a simple principle: the container must not introduce contamination, and it must not allow leakage. For heat- and moisture-sensitive medical devices like plastic Urine Sample Containers, ethylene oxide (EO) sterilization is the gold standard. EO sterilization is one of the few sterilization methods available for the sterilization of medical devises, and is essential for the protection of life saving medical applications.
The following are reasons EO sterilization is essential for the Urine Sample Container and the testing of infectious diseases:
1. The EO process operates at a maximum of 65° C, which means the EO process will not damage the polypropylene containers by causing warping or degradation, as can occur with other sterilization methods.
2. EO sterilization is capable of deep penetration and is able to thoroughly sterilize the entire contents of a container, and is able to, for example, sterilize the internal surfaces of an intricate and screw-capped container.
3. EO sterilization is designed to sterilize containers with capped tops as that integrated and engineered construction of components consists of post sterilization assembly.
4. EO sterilization is the only process that meets the Latin American standard for sterilization ISO 11135, ensuring the development and control of a process based on sterilization.
The container at the destination is completely sterile and is designed for the sensitive test.

With Regards to 2026 Regulations and Market Expectations
The global urine collector market is expected to reach $1.44 B in 2026 at a 9.7% CAGR with the growth in hospitalization, infection control, and chronic kidney disease. At this same time the regulations around the sterile transportation of medical devices is tightening.
ISO is currently developing ISO/CD TS 20327, a new technical specification providing requirements for handling, transporting, and distributing packaged sterile medical devices under the control of healthcare facilities.
For procurement teams evaluating Urine Sample Containers, EO sterilization capability is no longer optional. It is a compliance necessity.
•Alignment with WHO transport guidance: The World Health Organization’s latest infectious substance transport regulations require proper classification, packaging, marking, and labeling. EO-sterilized containers meet these rigorous requirements.
•Responding to evolving ISO standards: The latest changes to ISO 11135 and ISO 10993-7 have tightened EO allowances and increased requirements regarding validation of cycle EO consistency. Manufacturers must demonstrate that residual EO can withstand the new restrictions of toxicological testing through robust proof.
•Supply chain confidence: The EPA's 2026 draft of changed emissions restrictions suggests that EO sterilization must occur for medical devices to be available, making it necessary for healthcare practices to obtain supplies from compliant, validated manufacturers.
Trustlab EO-sterilizable Urine Sample Containers have been designed with these requirements in mind.
Protection by Design from Collection to Analysis
Trustlab really gets it – there should be no questions regarding safety in Urine Sample Containers, and we have fully integrated safety in our design with the GT208-517-1 (60ml) and GT208-517-2 (graduated 40ml):
•Closure with PE construction of the tightly fitting screw cap: Fulfills all exposure risk mitigation requirements by conducting leak-proof transport and securing the sample through the integrity of the entire logistics chain.
•Body in transparent PP construction with modulation of clear graduations: Fulfills the need to reduce risk by limiting access to potentially infectious specimens with the efficient and effective visual verification of volume.
•Polypropylene construction of a durable and chemical resistant design: Fulfills all requirements to withstand the strain of automated processing in the laboratory while fully compatible with EO sterilization processing.
•Lightweight construction for optimized transport: Fulfills all requirements in support of total high-volume logistics throughout clinical networks.
•EO sterilization for the heightened hygiene option: Fulfills all requirements for practitioners and healthcare establishments that handle infectious substances to the greatest assured sterilization level.
Trustlab’s Urine Sample Containers have many advantages to be used for routine urinalysis at clinical laboratories, microbiology sample collection and pathogen analysis, drug testing and toxicology research, and for transport of urine samples from collection points to laboratories.

Safety Meets Sustainability
By 2026, health care providers will use more sustainable bioprocessing materials for new medical devices and consumables. Hospitals are using bio-sourced polymers and bio-plastics. These materials reduce plastic waste and are safe for patient care.
TrustLab ensures Urine Sample Containers are constructed with sustainable materials and provides numerous benefits for healthcare facilities.
•Recyclable architecture: Containers are constructed with recyclable Polypropylene (PP) aiding healthcare’s circular economy and waste management goals.
•Infallible design: Containers are designed for optimized durability resulting in less waste.
•Optimized logistics: GT208-517-1 has a pack quantity of 500 with dimensions 58x34x43cm; GT208-517-2 has a pack quantity of 800 with dimensions 50x35x45cm. These pack configurations reduce the storage footprint and transport emissions.
Why TrustLab
TrustLab constructs its Urine Sample Container designs with intentionality. TrustLab is in continual alignment with global standards for EO-sterilizable lines of products to help encourage use and care for these materials.
Selecting TrustLab EO-sterilizable Urine Sample Containers is selecting a responsible transport container regarding pathogens, designed for safety, and diagnostics compliance.
Ready to enhance your specimen collection system? Reach out to our team for a quote, technical documents, sample requests, etc.
Trustlab—Engineering reliability for the laboratories of tomorrow.
FAQs
Q: Are Trustlab's EO-sterilizable Urine Sample Containers designed for routine urinalysis analyzers?
A: Yes. GT208-517-1 and GT208-517-2 are provided in standard dimensions and a clear body for easy integration with automated laboratory systems.
Q: What is the shelf life for EO-sterilized Urine Sample Containers?
A: Under the correct and recommended storage conditions in original, unopened packaging, containers are guaranteed to stay in a state of sterility for 24 months.
Q: Would EO-sterilized containers leave harmful residues on the samples?
A: No. Trustlab does validate EO sterilization cycles to meet the ISO 10993-7, confirming that the residual EO is well below the poison levels and safe for diagnostics.
Q: Is it possible to purchase non-sterile samples of the same Urine Sample Containers?
A: Yes. GT208-517-1 and GT208-517-2 can be bought in non-sterile versions. For those that do not need EO, sterilization is optional.
Q: How can I get samples and/or request a bulk quote?
A: You can initiate your request by selecting “GET FREE QUOTE” on this webpage, or you can reach out to our sales team. We aim to get back to you in 24 hours with all the information on pricing, specifications, and samples.